As an HTA Designated Individual you feel the responsibilities and challenges for compliance with the Human Tissue Act every day. You not only have an ethical and moral duty to comply but also a legal one. As an external HTA inspection nears, the question of whether you are HTA compliant may only increase your feelings of pressure. Especially if you do not have the required information readily available but instead rely on your researchers’ memories and notes.
You may have a team of Persons Designate who provide you with information for a Human Tissue Authority audit. However, without a standardised way of managing this information, it can take hours to manipulate into a usable format.
How much time would you save if you did not have to chase and pull together information prior to an audit? How much money would you save?
What else would you be working on if you were not spending time doing this administrative work? How much would this benefit your organisation in terms of your reputation, achievements and services offered as well as financially?
Using a laboratory sample management system to help your lab become HTA compliant
As HTA Designated Individuals and Licence Holders a Laboratory Sample Information Management System can offer you many benefits including:
- Reducing your administrative overheads when preparing for an HTA audit
- Removing your reliance on researchers’ memories to evidence compliance
- Increasing your time available to spend on strategic tasks
In addition, Sample Management Software can also help your team including your lab staff, researchers, Persons Designate and oversight officers. This can be achieved without increasing their workload.
Some Laboratory Sample Management systems, such as Achiever Medical, provide ‘silent’ auditing and compliance tools. These prompt for and automatically capture information as part of the in-built processes as users carry out their daily operations.
Further, lab storage management functionality provides visibility across your sample inventory and eliminates reliance on memory, post-it notes or spreadsheets.
Managing participant informed consent
Managing participant informed consent is a fundamental component of compliance with the Human Tissue Act. Advanced Sample Management Software, including Achiever Medical, provides comprehensive patient informed consent management. The software prompts users to enter informed consent and highlights any participants missing informed consent records.
However, equally as important is managing the withdrawal of informed consent. For some this can prove challenging.
If a participant withdraws consent do you know where all their samples are? What if the samples have been split into aliquots or used to create derivatives?
What if their sample was included as part of a Tissue Microarray?
To effectively manage this process, the software requires searchable biological sample family tree and Tissue Microarray structures. Within Achiever Medical, this information is used to identify and present all appropriate biological materials to the user. These can then be located and destroyed or returned in line with your SOPs.
HTA compliance insights
In addition to providing systems to assist compliance, displaying this information in an easily interpretable and accessible way is essential. Interactive, real-time dashboards can provide your Persons Designate with valuable insights at a glance. You can use them to highlight non-compliance as well as understand your current progress status. Therefore, you can arm your team with the information to address issues early and instil quality every day – not just in preparation for an audit.
The screenshot below displays the Oversight Officer’s interactive dashboards in Achiever Medical and how Persons Designate can use it to monitor HTA compliance. In addition, it can be used by those responsible for ensuring ethics approvals are in place.
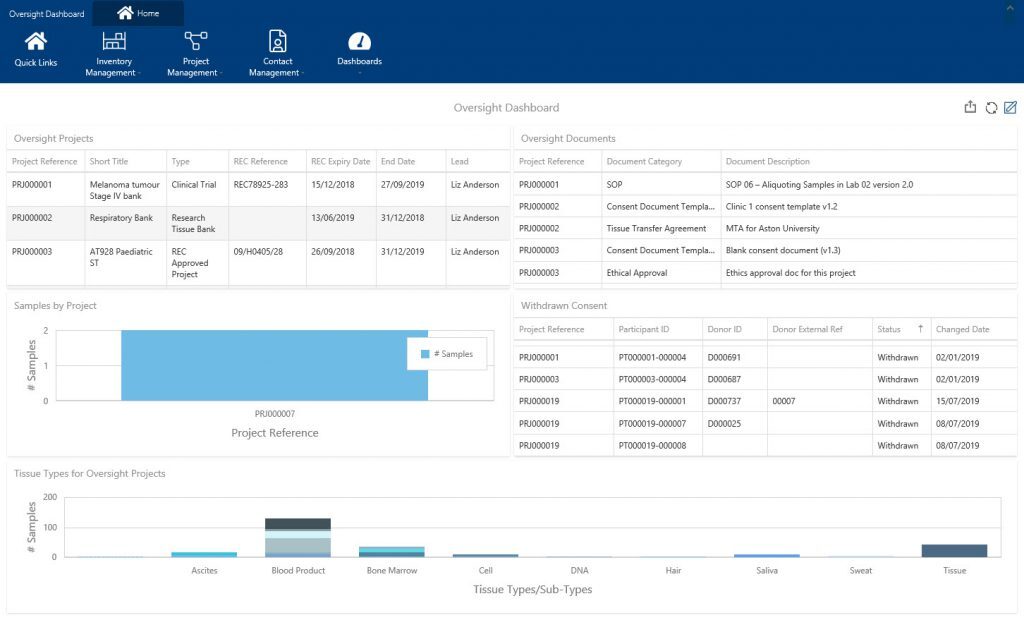
Final thoughts on helping your lab become HTA compliant
Your responsibility as an HTA Designated Individual and Licence Holder is fundamental to the continued operation of your institution and to make sure that your lab is HTA compliant. Compliance with the Human Tissue Act (HTA) is an ethical and legal requirement often requiring many weeks of preparation and late nights.
With the appropriate systems and processes in place to support you and your team, you can remove many of the administrative pressures of preparing for an HTA audit. Consequently, this can eliminate several layers of pressure and allow you to approach the audit with more confidence.
All of this is possible .
Chris Chambers, Leeds Teaching Hospitals NHS Trust says
“The University of Leeds chose to implement Achiever Medical to facilitate the safe and secure storage and management of human tissues used for research. It is in use on the main University campus and within the Leeds Teaching Hospitals Trust. We passed our HTA inspections in 2013 and 2017 and it was commented in the final report that the system enables good practice for tissue tracking. Achiever Medical has enabled multiple teams to move away from Excel-based record keeping to a robust database system, which in turn reduces potential for human error and loss of data. Removing manual audit tasks and the automation of repetitive data entry has created time for both clinicians and laboratory workers. This allows for more research and less concern about legislative compliance.”
Further reading
- Responsibilities and benefits of lab compliance’ – Responsibilities and Benefits of Laboratory Compliance


Comments are closed.