In this unprecedented and somewhat unsettling time, we’re seeing collaboration on a global scale like never before. On a human level, strangers are coming together to help each other through the health crisis. In business, instead of competing separately to find a cure companies are working together to target COVID-19. To quote Max De Pree, ‘We can accomplish more together than we can alone.’ Even under ‘normal’ circumstances, when it comes to clinical and scientific research, many studies and clinical trials involve collaboration between funders, labs, clinics, charities, industry and so on. And without collaboration with industry and researchers Biobanks and biorepositories would soon become obsolete. Collaborative Laboratory Information Management Systems (LIMS), like Achiever Medical, provide a range of tools to help you securely work together and, importantly, share information.
Managing multi-team projects, studies and clinical trials
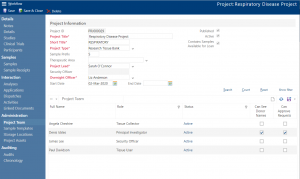
Achiever Medical’s unique project and study functionality enables you to segment and manage your samples as collections. Within each project you can link any associated Standard Operating Procedures (SOPs) and documentation. You can also allocate a project team. As you add each project team member you assign them a role. This role determines what data each person can see, enter and update. You’re in complete control. So, you can create projects that include individuals across multiple disciplines and teams to securely share specific samples and their data.
What’s more, within each project you can create studies. Each study has its own study team members with their assigned roles. This allows you to further segment your sample data and collections and gives you a more granular level of control. Plus, this allows you to elevate a user’s permissions for a specific subset of samples.
Rolling out the collaborative laboratory information management system to multiple teams and sites
Achiever Medical’s flexible configuration capability coupled with its unique permissions functionality means you can accommodate different teams’ process and data requirements in one system. Security controls protect personal identifiable information (PII) by encrypting data at source. You can even categorise samples differently across your different teams.
However, with all your samples in a single system your oversight officers and HTA licensees interrogate one application to check and confirm compliance. Plus, because Achiever Medical is web-based it’s easy for you to grant access to other sites and teams when you need. Finally, if you’re hosting the system on your network your IT team has just one system to manage.
“We have been using Achiever Medical to manage and segregate the data of separate teams for 9 years and this now extends to 31 teams. Achiever Medical has also been extended to managing our internal auditing and will allow us to record consent appropriately and manage/remove data as required, to fully comply with GDPR.” Chris Chambers, Leeds Teaching Hospitals NHS Trust
Sharing samples – internally and externally
Sharing resources helps you get maximum value from them. And it’s no different when it comes to samples – depending on consent restrictions, naturally. If you’re able to reuse and repurpose samples, by using Achiever Medical you could make these available to researchers within your own institute or even externally.
As a physical or virtual Biobank, it’s essential for you to be able to publicise and share your samples. Similarly, if you’re a pharmaceutical company you may want to be able to share your samples between your clinical and discovery teams – patient and study consent permitting.
Achiever Medical’s sample request process that makes it easy for you to highlight samples that are available for loan. Researchers can securely search through your samples to request them. You can then approve or reject requests based on their intended use. You’re in complete control of which external researchers you give access to and you decide what information is available to them. Requesters can keep track of their requests and Achiever Medical’s dispatch process helps you track shipments. Plus, the system automatically maintains the chain of custody.
Hosted systems for specific clinical trials and disease-specific studies
Bringing a fully functional LIMS online as and when you need it might seem impossible. However, hosted systems like Achiever Medical can make this a reality.
Thanks to its underlying technology as well as its project and study management capability, you can securely and quickly allocate access and permissions to authorised users within your institute and your collaboration partners.
Plus, Achiever Medical’s disease management module enables you to capture and analyse disease-specific data. Each disease has its own characteristics, parameters and data set. Within the system you can define each disease and choose exactly what data you want to capture including setting any lookup options and mandatory information.
The system’s bulk data import tools allow authorised users to upload data in a consistent format. In addition, Achiever Medical’s query and dashboard tools allow you to easily analyse your data. Plus, your authorised users can securely export data at the end of the study.
A final thought about collaborative Laboratory Information Management System
Collaboration is essential to the success of Biobanks and clinical research. Some collaborations exist for short periods of time, while others span years. Successfully sharing resources and information are fundamental to successful collaborations.
Having the right underpinning technology and software can facilitate collaboration and information sharing. Saving you time, improving efficiency, promoting consistency, increasing quality and maximising resources. A modern web-based laboratory information management system that support you working collaboratively can make this a reality. Ultimately helping you ‘Make Every Sample Matter’.


Comments are closed.