With the virus increasing amongst young people, Universities are focusing on what additional measures they need to put in place to safeguard the health and well-being of staff and students returning to campuses and halls of residence. Many Universities with in-house lab facilities are considering, and even starting to conduct, regular, random COVID-19 testing for students and staff. Hoping to successfully identify any COVID-19 positive, asymptomatic individuals, so they can rapidly notify them to halt further spread. Preventing any potential outbreaks is time-sensitive requiring quick and accurate testing as well as prompt action.
So, for any in-house COVID-19 testing of University students and staff to be successful you need to process samples quickly – and at scale. But you also need an effective communications process in place to notify those impacted. All of which rely on you having the right systems in place to support you.
Achiever Medical is a secure, web-based Laboratory Information Management System (LIMS) that can help you to securely and efficiently manage, process and track thousands of samples. What’s more, its integrated donor profiling and customer relationship management (CRM) capabilities enable you to send targeted communications.
Capturing and storing samples for testing
Whether you’re registering samples by bulk uploading a CSV file or Excel spreadsheet, receiving them using a handheld scanner or manually entering the data, Achiever Medical LIMS formats and standardises your data to make sure it’s consistent. If you’re not carrying out the tests immediately and want to store the samples, storage management in the solution enables you to define your storage containers and track sample movement.
If you’re handling human tissue samples in the UK, then compliance with the Human Tissue Act (HTA) is essential. So, it’s important that you have the appropriate level of consent for any work you’re doing with the samples. Especially if you’re planning on aliquoting and storing them for future studies. Achiever Medical LIMS enables you to record and track informed consent alongside the student or staff member. This includes managing opt-ins and handling consent withdrawals. Plus, all sample events, such as aliquoting and analysing, are audited and the sample genealogy captured for complete traceability.
Protecting sensitive and personal identifiable information (PII)
You can manually enter or bulk import test results into Achiever Medical LIMS.
Your University comms teams needs to communicate outcomes and required actions to those impacted by a positive test. And this needs to be done quickly and sensitively. With Achiever Medical LIMS’ data protection and inbuilt ‘at-rest’ encryption you can give your University comms team access to individuals’ contact data while protecting this from researchers carrying out the tests.
What’s more, you can choose how to communicate with affected individuals within Achiever Medical LIMS. This might be via email or SMS or both. Plus, you can send targeted communications to those living in the same halls as someone who’s tested positive. Also, you can automate any communications based on predefined rules. For example, the system sends an automated email only once you’ve verified the results.
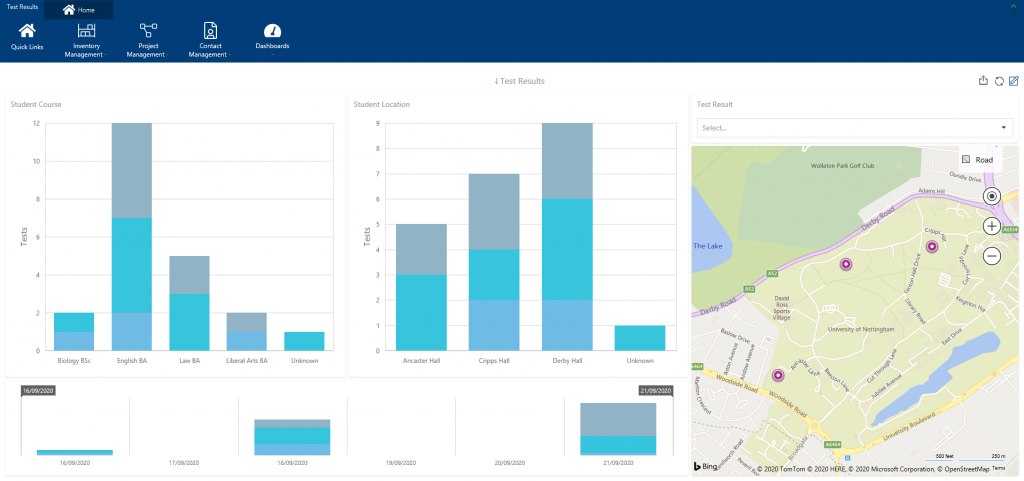
Interactive dashboards highlight hotspots
Having oversight of where the major outbreaks are across your University campus can help you identify other areas that may require your attention.
Interactive dashboards allow you to focus on specific locations, characteristics and track progress over selected time-periods. You can also drill into the underlying data for closer inspection.
Time is of the essence
When it comes to Universities testing students and staff for COVID-19 quick turnaround times make a difference. Likewise, when it comes to deploying a system to support your testing efforts.
Achiever Medical LIMS is available as an on-premise or hosted solution. The solution also includes a set of standard sample processing workflows as well as a library of dashboards. This allows your labs and Biobank to start using the system within weeks. While its configuration tools allow you to enhance and extend the system as and when required.
Achiever Medical LIMS is project- and study- centric which means you can easily extend the system to manage and process non-COVID-related samples and diseases. Giving you a system that supports your sample management requirements for the longer-term.
Download the information sheet and Get in touch for more information and to see how Achiever Medical LIMS can help Universities manage onsite testing of students and staff for COVID-19.


Comments are closed.